 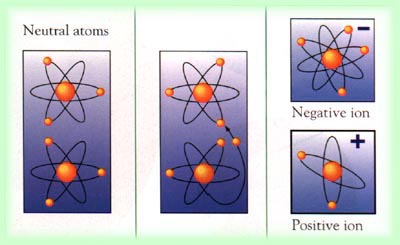 
The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. When many positive (+ve) and negative (-ve) ions come together. A cation is a positively charged ion with fewer electrons than protons while an anion is negatively charged with more electrons than protons. The atom becomes either a positively or negatively charged particle called an ION. Many positive (+ve) and negative (-ve) ions arrange themselves in a structure called a crystal. What is an ion vs anion An ion (/an, -n/) is a particle, atom or molecule with a net electrical charge. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. When an atom loses or gains an electron, an imbalance occurs. Write the electron configuration of oxygen atom (Z=8). In a semiconductor diode, the two bias conditions are ). (a) neutral zone (b) recombination region (e) depletion region (d) diffusion area 2. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. In a semiconductor diode, the region near the pn junction consisting of positive and negative ions is called the ). An atom that loses an electron forms a positively charged ion called a cation an atom that gains an electron forms a negatively charged ion, called an. When they do, they become monatomic ions. Individual atoms can gain or lose electrons. 
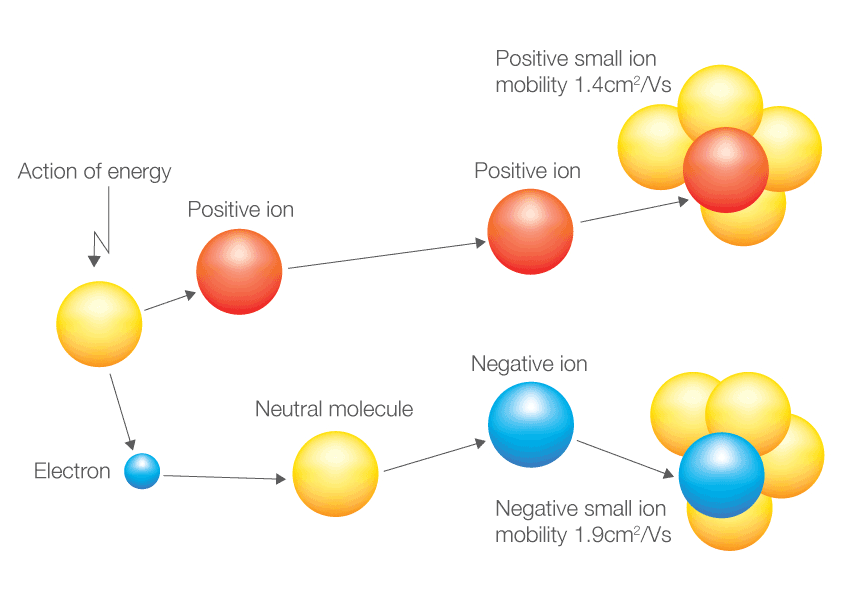
The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. The atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called an anion). Compounds formed from positive and negative ions are called. Notice that there are no leftover electrons. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. For example, group 17 elements (one group left of the noble gases) form 1 ions group 16 elements (two groups left) form 2 ions, and so on. On the right, the chloride ion has 18 electrons and has a 1− charge. Moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble gases. On the left, the chlorine atom has 17 electrons. Generally, positive ions weigh more than negatively charged ions, and mobility values are larger than 0.4-0.8 (cm2/Vs). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
